Step 1: Cell/Tissue Homogenization

![]()
The sample is homogenized in an isolation buffer that is highly chaotropic to facilitate effortless and complete solubilization. The SPLIT for Blood protocol adds a red blood cell lysis step prior to sample homogenization.
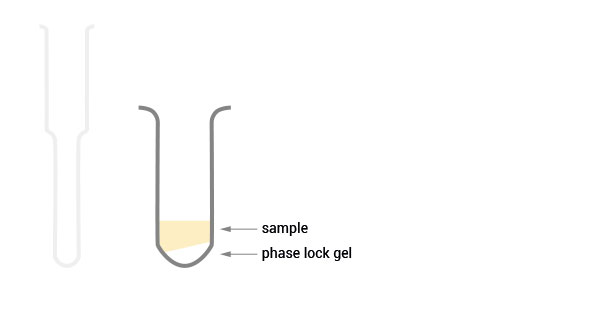
The sample is transferred to a phase lock gel column which contains a special gel matrix that acts as a barrier between the organic and aqueous phase based on the density differences.
Step 2: Phenol Extraction
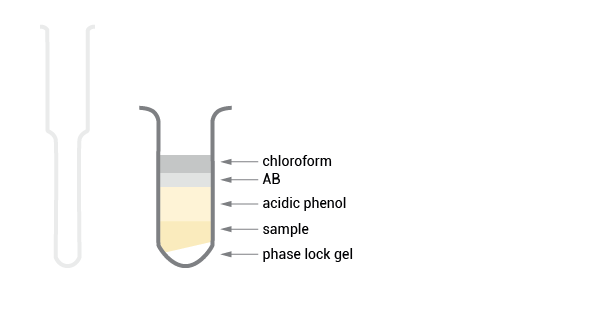
Acidic phenol and acidic buffer are added to create a monophasic solution, which is essential for the efficient separation of genomic DNA into the organic phase. Chloroform is added and phases are cleanly separated.
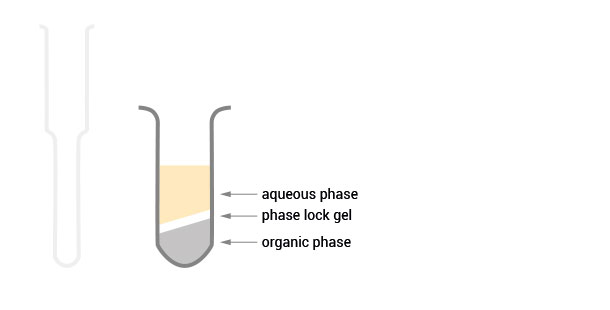
After centrifugation the gel acts as a seal between the phases.
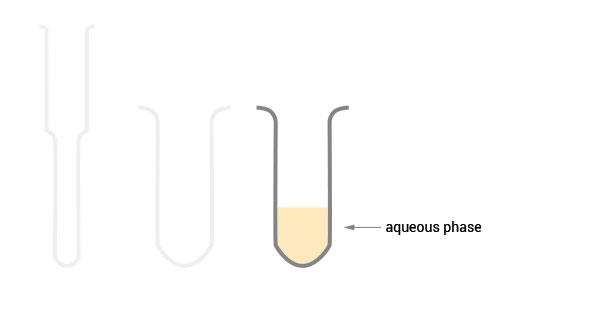
The aqueous phase containing the RNA can be easily decanted and no carry-over of the organic phase will take place.
Step 3: Purification of RNA Fractions
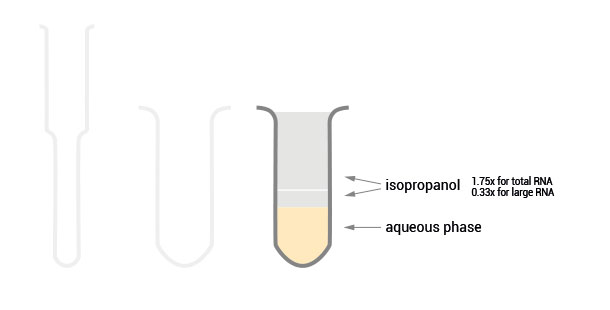
Depending on the amount of isopropanol added either the total RNA (1.75x isopropanol) can be extracted or the RNA can be split in a large (0.33x isopropanol) and a small RNA fraction (1x isopropanol to the flow-through of the large RNA fraction).
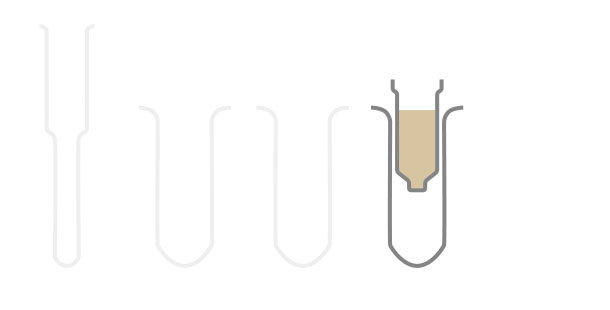
The RNA is precipitated onto a silica column. For the total RNA the entire RNA will precipitate onto the silica carrier, while for the large fraction RNA with a lower limit of about 150 nt will bind whereas the small RNA will be in the flow-through.
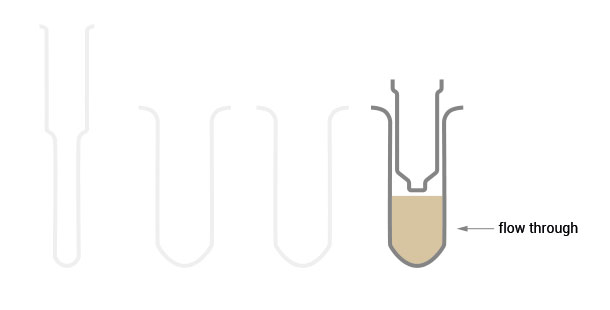
The flow-through of the large RNA fraction contains the small RNAs which didn’t bind to the silica column. After addition of 1x isopropanol it can be precipitated and purified onto a new silica column.
Step 4: Elution of RNA
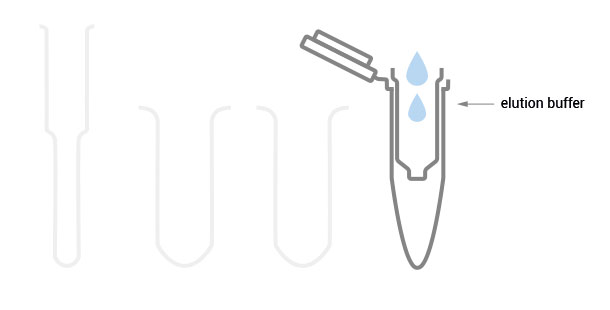
10 – 50 µl of Elution Buffer or Storage Buffer are added to the silica membrane to elute the RNA bound to the silica carrier.
RNA extraction is finished and the RNA is ready for any downstream application.



